New Viral Vector for Improved Gene Therapy in Sickle Cell Disease
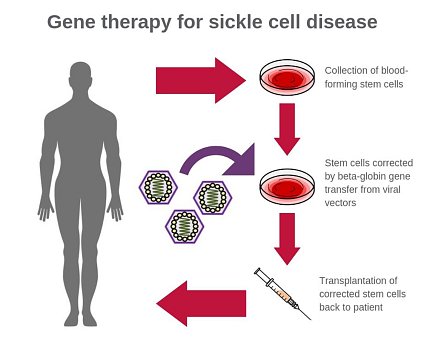
Researchers at NIH have developed a new and improved viral vector—a virus-based vehicle that delivers therapeutic genes—for use in gene therapy for sickle cell disease. In advanced lab tests using animal models, the new vector was up to 10 times more efficient at incorporating corrective genes into bone marrow stem cells than the conventional vectors currently used, and it had a carrying capacity of up to 6 times higher, the researchers report.
The development of the vector could make gene therapy for sickle cell disease much more effective and pave the way for wider use of it as a curative approach for the painful, life-threatening blood disorder. Sickle cell disease affects about 100,000 people in the United States and millions worldwide.
“Our new vector is an important breakthrough in the field of gene therapy for sickle cell disease,” said study senior author Dr. John Tisdale, chief of NHLBI’s Cellular and Molecular Therapeutic Branch. “It’s the new kid on the block and represents a substantial improvement in our ability to produce high capacity, high efficiency vectors for treating this devastating disorder.”
The new vector, for which NIH holds the patent, still needs to undergo clinical testing in humans.
The study was supported by NHLBI and NIDDK and was published online Oct. 2 in Nature Communications.
